|
It is important to note how the location of elements on this table tells us about their properties. From this deduction, he formed the periodic table. In 1869 Russian chemist Dmitri Mendeleev noticed there existed an innate pattern of organization for the chemical elements. Meanwhile, elements in the same period have the same number of occupied electron shells. Elements in the same group have the same number of valence electrons. The periodic table is organized into groups (vertical columns), periods (horizontal rows), and families (groups of elements that are similar). You can view all sorts of trends, properties, magnetism, electrons, and even articles on all the elements! The Periodic Table and the Periodic Trends We think our periodic table is one of the best in the world! Visit our new interactive periodic table. View trends, families & groups interactively! Families: Elements that have the same number of valence electrons and therefore similar properties.Periods: The horizontal rows in the periodic table that signify the number of electron shells in an element.Groups: The vertical column of the periodic table that signifies the number of valence electrons in an element.Elements: A pure substance composed of a single atom with a unique atomic number.How to Write Electron Shell Configurations.If you enjoy this article, be sure to check out our others! Related Articles: In addition, you will learn about the different properties of the periodic table groups, periods, and families. We will take a close look at the groups of the periodic table. (3) It's a lot easier to remember facts about 18 elements than over 100 elements.In this tutorial, you will learn how to read the periodic table. (2) These eighteen elements make up most of the matter in the Universe. Remember that the orbitals are the places you will generally find the electrons as they spin around the nucleus. (1) Electrons fit nicely into three orbitals. Since the launch of the site, we've been asked, "Why start with 18?" The rules for the first eighteen elements are very straightforward: You will never stop discovering new reactions and compounds, but the elements will be the same. With the tools you learn here, you can explore and understand the Universe. The iron atoms in the red soil of Mars are also the same. Iron (Fe) atoms found on Earth are identical to iron atoms found on meteorites. While there are more elements to discover, the basic elements remain the same. 
Scientists just confirmed the creation of element 117 in 2014. Up to this point in time, we have discovered or created about 120. 
Chemists have learned that over 95% of your body is made up of hydrogen (H), carbon (C), nitrogen, oxygen, phosphorus (P), and calcium (Ca).Īs far as we know, there are a limited number of basic elements. For example, you are made up of billions of billions of atoms but you probably won't find more than 40 elements (types of atoms) in your body. The term 'element' is used to describe atoms with specific characteristics. 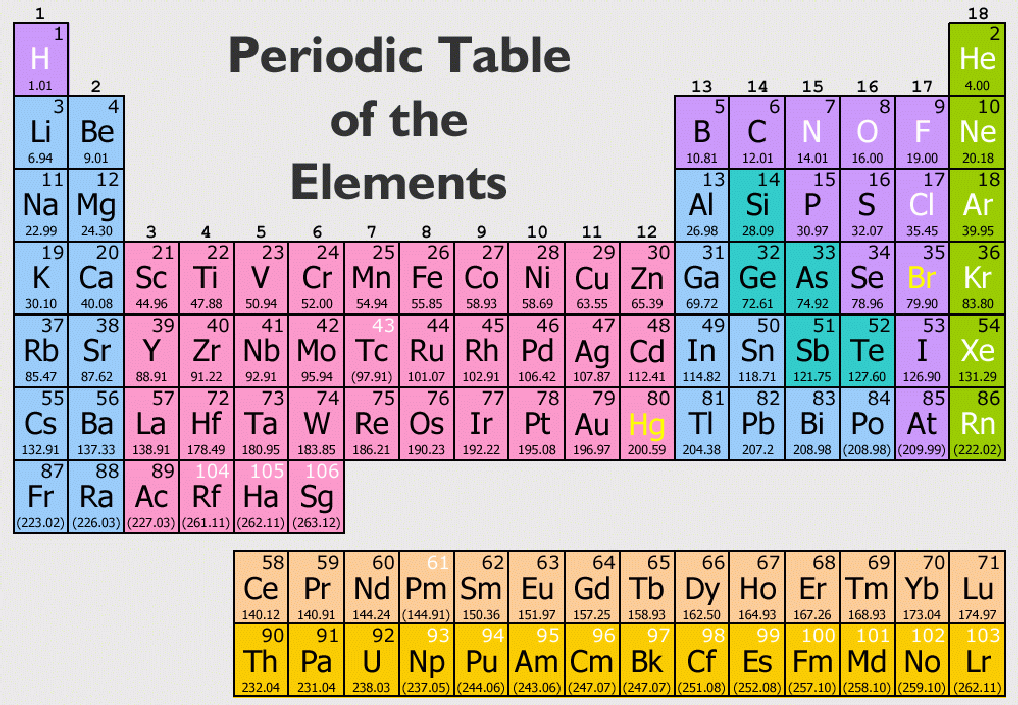
Remember that 'atom' is the general term. The atoms for each element are unique, even though they are all made of similar subatomic parts. If you have seven protons, neutrons, and electrons, you will have a nitrogen (N) atom. If you have eight protons, neutrons and electrons, you will have an oxygen (O) atom. When those pieces start combining in specific numbers, you can build atoms with recognizable traits. You know that a generic atom has some protons and neutrons in the nucleus and some electrons zipping around in orbitals. Now we're getting to the heart and soul of the way the Universe works.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
